
Mitochondrial quality control and neurodegenerative diseases
Our laboratory focuses on understanding the roles of mitochondrial quality control and metabolism in neurodegenerative diseases. We use unbiased proteomics and genomics, cellular and molecular biology, patient iPS cells-derived cell culture and diseased animals to investigate the fundamental mechanisms by which mitochondrial impairment contributes to cellular metabolism disturbance and neurodegeneration. We also utilize rationally designed peptide inhibitors of protein-protein interactions and the high throughput screening approach to developing “mitochondrial medicine” as therapeutic strategies for treating neurodegenerative diseases.
Mitochondria are essential organelles that support cellular function by regulating energy metabolism, generating ATP, and maintaining calcium homeostasis. When mitochondria become dysfunctional, they produce excessive reactive oxygen species and exhibit impaired metabolic activity, which disrupts key biological processes, including cellular bioenergetics, immune responses, genomic stability, and programmed cell death.
To mitigate these detrimental effects, mitochondria employ several quality control mechanisms that preserve their multifaceted functions and alleviate mitochondrial stress. These quality control pathways include mitochondrial dynamics (fusion and fission), mitochondrial proteostasis, and communication with other subcellular organelles. Together, these processes promote the repair of damaged mitochondrial components, facilitate adaptation to stress, and eliminate irreversibly damaged mitochondria.
Our research focuses on elucidating the roles of mitochondrial quality control-related proteins in maintaining mitochondrial and cellular function, particularly in the context of neurodegenerative diseases. Using a series of inhibitors targeting aberrant mitochondrial pathways, we investigate whether modulating mitochondrial quality control offers a viable therapeutic strategy. In parallel, we apply unbiased proteomic and genomic approaches to identify key regulators of mitochondrial proteostasis and organelle communication, aiming to uncover how mitochondrial protein and genome homeostasis govern cell survival and contribute to the progression of neurodegeneration.
Mitochondrial Quality Control Mechanism and Neurodegeneration
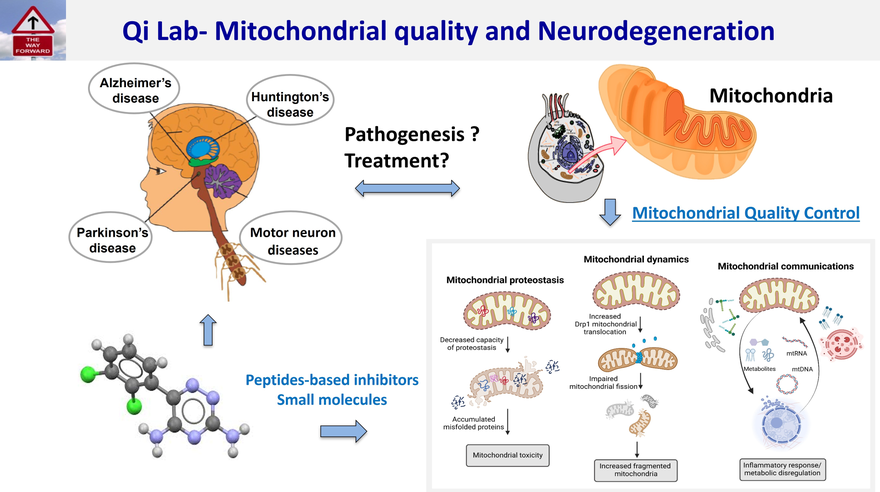
Mitochondrial quality control includes mitochondrial dynamics (fusion/fission), mitochondrial protein homeostasis, and mitochondria-associated organelle communications. These events maintain mitochondrial health and cell survival. Impairment of the mitochondrial quality control system contributes to neurodegeneration.
| Major Research Areas |
|---|
| Cell Death Mechanisms, Mitochondrial biology and pathology, Neurodegenerative diseases, Therapeutics |
| Disciplines |
| Drug discovery , Metabolism, Molecular and cellular biology, Neuodegenerative diseases , Neuroscience, Pharmacology and Therapeutics, Signal Transduction |
| Organ Systems |
| Nervous System |
| Diseases |
| Alzheimer's Disease, Huntington's Disease, Metabolic syndrome , Neurodegenerative Diseases, Neurological Disorders, Parkinson's Disease |
| Major Techniques |
| Cell Culture, Cellular and Molecular Biology, In-vivo Animal Models, Induced Pluripotent Stem Cells (iPSCs) from Patients, Peptide Drug Design |