
Molecular mechanisms of membrane remodeling, fission and fusion in endocytosis and mitochondrial dynamics
Cellular membranes undergo dynamic changes in shape in remarkably striking ways during processes such as cell movement and division, and also during vesicle fission and fusion events that are integral to intracellular membrane trafficking. Large, mechanochemical GTPases of the dynamin superfamily are critically involved in many of these events including those of synaptic vesicle recycling, clathrin-mediated endocytosis, mitochondrial division and fusion, and peroxisome biogenesis. They often work in concert with membrane-sculpting BAR domain-containing proteins and various accessory molecules in effecting membrane fission and fusion. The main objective of our research is to reconstitute and elucidate the various molecular machineries and mechanisms involved in intracellular membrane remodeling, fusion and fission, specifically in synaptic vesicle endocytosis and mitochondrial dynamics, using cutting-edge state-of-the-art fluorescence spectroscopic techniques, including FRET, FCS, FLIM and related biophysical methodologies.
Dynamin-dependent Endocytic Vesicle Scission
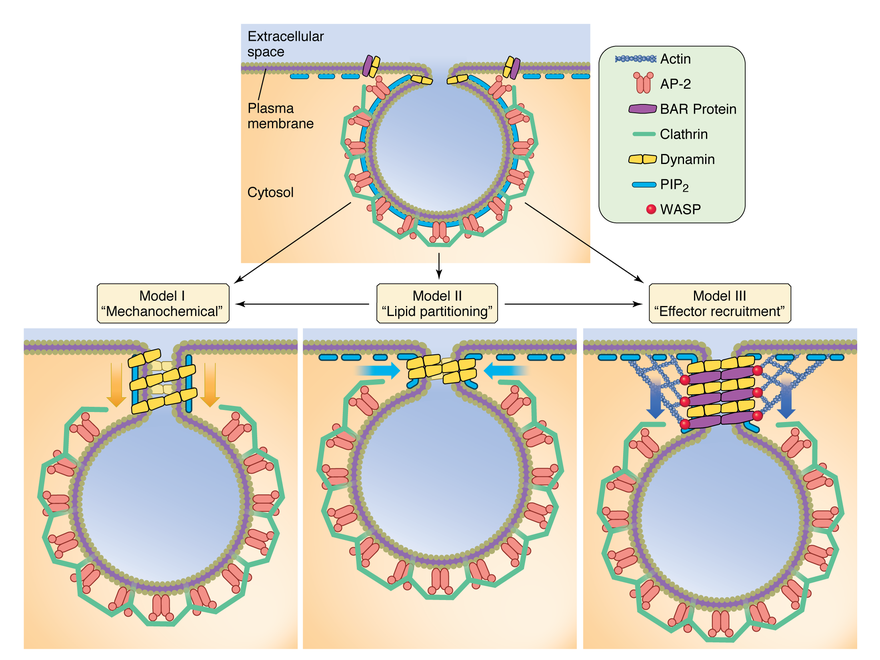
Models for dynamin-dependent clathrin-coated vesicle formation. In model I, dynamin undergoes a nucleotide-dependent conformational change that either constricts, stretches or twists the underlying membrane tubule to mediate membrane fission. In model II, dynamin in the assembled and/or constricted state clusters PIP2 leading to membrane destabilization and fission. In model III, dynamin co-assembles with membrane curvature-generating BAR proteins (e.g. SNX9, syndapin) that function cooperatively with the actin network to drive membrane fission and vesicle release.
| Major Research Areas |
|---|
| Membrane Fission, Membrane Trafficking, Mitochondrial Function, Protein Folding, Protein Structure/Function, Protein-Protein Interactions |
| Disciplines |
| Biophysics, Fluorescence Spectroscopy, Protein Folding |
| Organ Systems |
| Cardiovascular System, Nervous System |
| Diseases |
| Cancer, Heart Failure, Neurodegenerative Diseases |
| Major Techniques |
| Confocal Microscopy, Stopped Flow Spectroscopy |