
Neurotransmitter release evoked by sympathetic activation.
Varied sympathetic activity is translated into differential hormonal profiles under the sympatho-adrenal stress response. Our lab studies multiple components of this vital physiological response system. We study the synaptic peptide transmitter regulation of hormone release at the splanchnic-adrenal synapse and we study the regulation of activity-dependent differential transmitter release from neuroendocrine adrenal medullary chromaffin cells. We also have a project in collaboration with the UCLA Cardiac Arrythmia Center developing technologies for the measure of neurotransmitters released under sympathetic activation in the heart.
Activity-dependent differential transmitter release.
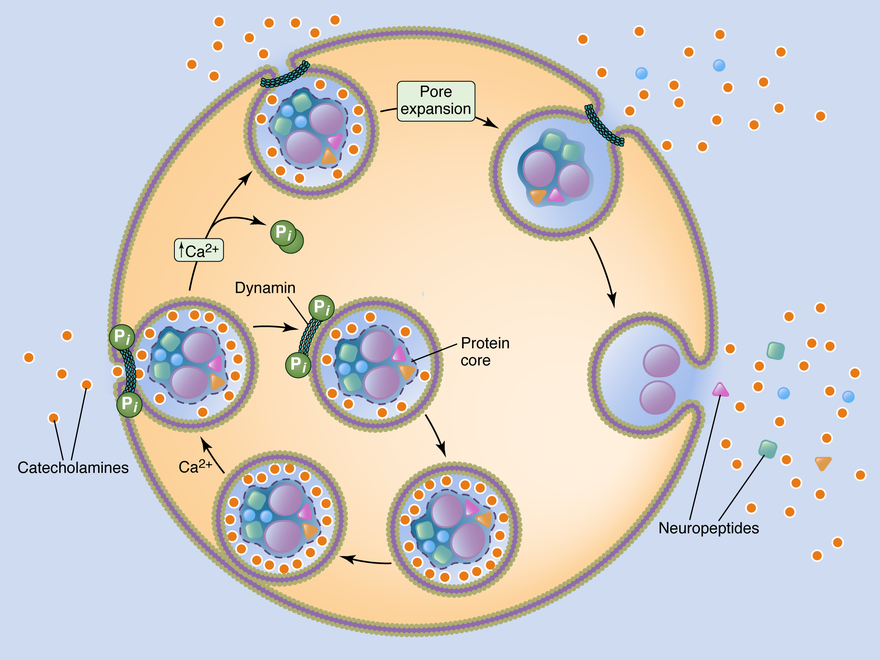
A cartoon representation of the shift in the secretory mode from neuroendocrine adrenal chromaffin cells. At basal stimulation, as under sympathetic tone, a sub-fraction of soluble catecholamine is released through a restricted fusion pore. This mode of exocytosis is termed "kiss and run", referring to the omega-like profile of the fused granule. Under elevated stimulation, as under the sympatho-adrenal stress reflex, the fusion pore is driven to expand, collapsing the granule into the cell surface. This "full collapse" mode of secretion facilitates maximal release of catecholamine as well as the proteinaceous peptide transmitter-containing granule core.
PACAP as a primary effector of the sympatho-adrenal stress response.
Environmental insult, injury, illness and psychological trauma all evoke the “fight or flight” sympathetic stress response. Specific stressors selectively evoke elevations in serum epinephrine (Epi) or norepinephrine (NE) to formulate the appropriate physiological response. For example, hypoglycemia leads to selective Epi release to increase hepatic blood flow, increase glucagon secretion and to decrease insulin sensitivity, all in order to elevate blood glucose levels. Acute cold stress results in selective NE release to constrict peripheral vasculature in order to preserve body heat. Hypoxia results in an equivalent increase in both Epi and NE to increase cardiac output and pulmonary function to deliver oxygen to the tissues of the body. Norepinephrine is released from sympathetic nerve terminals throughout the periphery, as well as from neuroendocrine chromaffin cells of the adrenal medulla while the adrenal medullary chromaffin cells are the sole source for serum Epi. There are two secretory isotypes of adrenal chromaffin cell; Epi-secreting or NE-secreting. Thus, differential NE versus Epi release dictates a stressor-specific activation of the adrenal medullary Epi-secreting chromaffin cells. Adrenal chromaffin cells receive synaptic input from the sympathetic splanchnic nerve. Previous studies have shown that the splanchnic synapse utilizes a dual transmitter excitation system. Under basal sympathetic tone, the splanchnic terminals release rapidly acting and deactivating acetylcholine to fine-tune adrenal excitation. Under this “rest and digest” condition, the sympathetic nervous systems works in concert with the parasympathetic nervous system to regulate homeostatic processes. Stress evokes increased sympathetic drive and signals persistent and robust adrenal excitation through splanchnic release of the potent chromaffin cell secretagogue, Pituitary Adenylate Cyclase Activating Peptide (PACAP). Thus, splanchnic PACAP is the stress transducer in the sympatho-adrenal stress reflex. Despite its physiological importance, relatively little is known of how splanchnic PACAP controls differential Epi versus NE release from the adrenal medulla.
Fast detection of catecholamines and neuropeptides in a beating heart.
The sympathetic nervous system modulates cardiac function by controlling key parameters such as chronotropy and inotropy. Sympathetic control of ventricular function occurs through extrinsic innervation arising from the stellate ganglia and thoracic sympathetic chain. In the healthy heart, sympathetic release of norepinephrine (NE) results in positive modulation of chronotropy, inotropy, and dromotropy, significantly increasing cardiac output. While norepinephrine (NE) is the primary neurotransmitter for these projections, neuropeptide Y (NPY) is an abundant cardiac co-transmitter. NPY plays a vital role in homeostatic processes such as angiogenesis, vasoconstriction, and cardiac remodeling. Elevated sympathetic stress, resulting in increased release of NE and NPY, has been implicated in the pathogenesis of several cardiovascular disorders including hypertension, myocardial infarction, heart failure and arrhythmias, which may result in sudden cardiac death. Methodologies for detection of NE and NPY in cardiac tissue are limited. Present techniques rely on microdialysis for analysis by high-performance liquid chromatography coupled to electrochemical detection (HPLC-ED), radioimmunoassay, or other immunoassays, such as enzyme-linked immunosorbent assay (ELISA). Although significant information about the release and action of NE and NPY has been obtained with these methodologies, they are limited in temporal resolution, require large sample volumes, and provide results with a significant delay after sample collection (hours to weeks). We have an active collaboration with the UCLA Cardiac Arrhythmia Center to develop and deploy novel approaches for measurement of interstitial cardiac norepinephrine, NPYU and other peptide transmitters, using minimally invasive, electrochemical approaches applied in a beating porcine heart. With these new tools, we have collected the first multispatial and high temporal resolution, multichannel in vivo measurements of NE, NPY and other peptide transmitters both in the myocardium and vascular space.
Activity-dependent release of adrenal peptide stress hormones.
Environmental threat, physical exertion or injury and psychological strain all lead to the initiation of the sympatho-adrenal “fight or flight” stress response. Neuroendocrine adrenal medullary chromaffin cells receive excitatory synaptic input from the sympathetic splanchnic nerve. Splanchnic activation causes adrenal chromaffin cells to release catecholamines as well as a diverse array of neuro- and vaso-active peptide transmitters into the circulation. Different levels of stress result in the differential release of catecholamine versus peptide transmitters to formulate the appropriate physiological response. All secreted hormones, catecholamines as well as multiple species of peptide transmitters, are co-packaged in the same secretory granules. Thus, differential hormone release is regulated at a step after granule fusion. Under basal chromaffin cell excitation, set by the sympathetic tone, selective release of freely-soluble catecholamine occurs through a transient fusion event, characterized by a narrow, structured exocytic fusion pore between the granule lumen and the extracellular space. Under sympathetic tone, selective and modest catecholamine release plays an important role in the “rest and digest” metabolic status of energy storage, regulating homeostatic physiological functions including pancreatic insulin secretion, increased blood flow to the viscera and maintenance of basal cardiac activity. In response to stress, increased chromaffin cell stimulation modulates the mode of secretory granule fusion, leading to the expansion of the exocytic fusion pore to maximize catecholamine release and facilitate exocytosis of the co-packaged adrenal peptide transmitters. Elevated serum catecholamine levels, in combination with adrenal-derived peptide transmitters, are core effectors of the sympathetic “fight or flight” stress response. Together they regulate multiple processes that prepare for defense or escape, including generalized analgesia (enkephalin), increased cardiac output (elevated catecholamines), blood flow to skeletal muscle (atrial natriuretic factor, Neuropeptide Y) and blood glucose (pancreastatin). Thus, regulated expansion of the secretory fusion pore represents a key element of the acute stress response.
| Major Research Areas |
|---|
| Autonomic Nervous System, Biochemical Pathways, Ion Channels, Membrane Trafficking, Synaptic Transmission |
| Disciplines |
| Electrophysiology, Molecular Biology, Signal Transduction |
| Organ Systems |
| Cardiovascular System, Endocrine System, Nervous System |
| Diseases |
| Autonomic Regulation, Cardiac Arrhythmias, Hypoxia, Stress |
| Major Techniques |
| Amperometry, Confocal Microscopy, Electrochemistry, Electrophysiology, Immunohistochemistry, Ion Transport, Patch Clamping, Protein Expression, Quantitative Fluorescence Imaging, RT-PCR |